ISO 13485:2016 certified & FDA registered
Our Aspiration
Medical instruments of highest quality and precision
Impeccable quality standards are part of our business philosophy and has been an integral part of our journey since 1932. Our premium quality “Made in Germany” distinguishes our manufactory regardless of an increasingly competitive market.
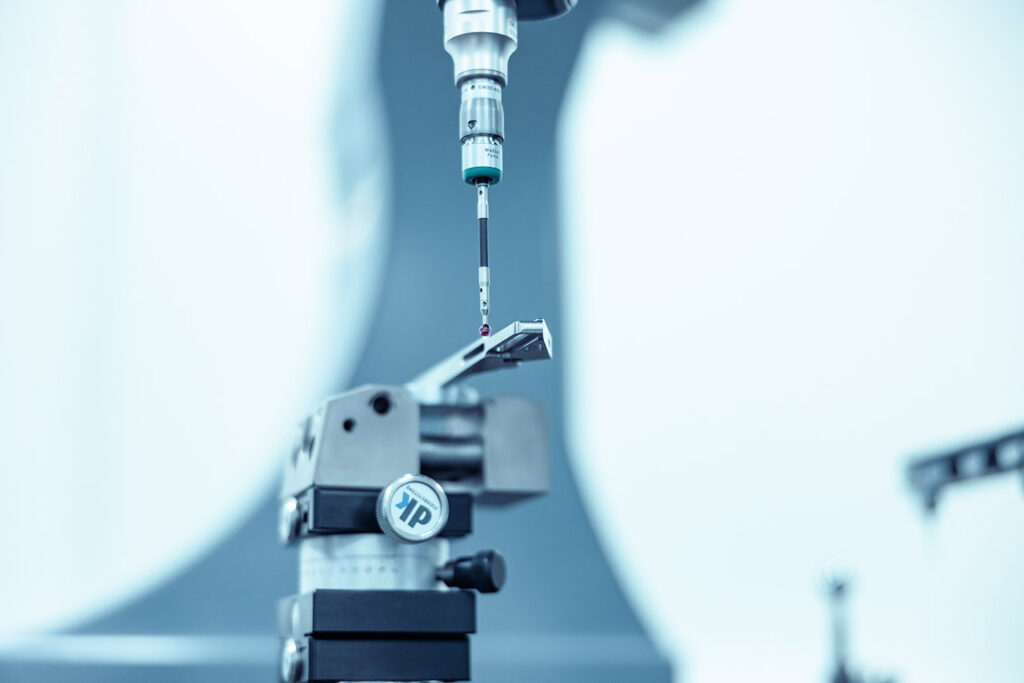
When using surgical instruments, the highest quality and precision is of utmost importance. In the realm of medical instruments especially, safety and the patients’ health sit at the very core of our work which we ensure constantly through our comprehensive quality management-system. Our quality management is certified ISO 13485:2016 and continually applied and refined by our employees. Procedural instructions, in-house standards based on the guidelines of ASTM International (American Society for Testing and Materials), qualified production equipment, and validated processes ensure that you can count on surgical instruments of the highest quality in both our RW Standard Range and our RW Indiviual Instruments.
In addition, specified quality control-measures such as the continuous monitoring of important process parameters, the regular monitoring of production units and, above all, in-process-monitoring guarantee the highest standards of quality. All of these aspects facilitate transparency and permanent traceability throughout the production processes.
As an FDA registered company, production is carried out according to FDA regulations CFR (Code of Federal Regulations) Title 21 Part 820. “Current good manufacturing” is practiced (CGMP).
Therefore, all relevant international requirements are met in order to be able to supply global markets with surgical instruments.